Other works performed at the IBR-2
- Review on role of Aβ peptide in development of Alzheimer’s disease published in Natural Science Review journal
- Most stable structural phase of biocompatible zirconium dioxide found
- FLNP JINR researchers evaluated effect of rare-earth metals on cyanobacteria
- Assessment of chemical elements in soil in Romania
- On development of semi-interpenetrating polymer-micellar networks
- Peculiarities of charge-discharge processes in PW electrodes with different level of dehydration
- Gamma radiation-induced alterations in nanocrystalline titanium nitride particles
- Neutron diagnostics of nuclear reactor surveillance specimens
- Neutron scattering helps in developing burn wound dressings with desired properties
- Authors of USPEX from SKOLTECH on a visit to FLNP. New superconducting hydrides: predict, synthesize, and study
- Neutron scattering, membrane biophysics, and Alzheimer's
- Galfenol: studies of alloys for «energy harvesting»
- Preserving history captured in granite
- Neutrons reveal secrets of shape memory alloys
- Fullerenes in the fight against amyloids: under scrutiny of neutrons
- Magnetic textiles for biocatalysis. Neutron investigations help to explain the structural features of a complex nanocomposite material.
Scientists from FLNP JINR in collaboration with their colleagues investigated the structural properties of cell membranes and the interactions occurring in them, which play an important role in the development of such devastating neurodegenerative diseases as Alzheimer's.
Alzheimer's disease – a threat to humanity
Alzheimer's disease, one of the most expensive diseases in terms of caregiving costs, ranks sixth among the leading causes of death and affects mainly the elderly. Devastating changes in the brain cause gradual loss of memory, disorientation, deepening confusion about events, time and place, and eventually lead to severe dementia. Today, Alzheimer's affects tens of millions of people worldwide, and by 2050, this number is expected to exceed 130 million. The disease, which is called the scourge of the developed countries of the 21st century, was named after German psychiatrist Alois Alzheimer, who first studied and described the symptoms and development of the disease a little over a century ago. But despite decades of research, the underlying causes and mechanisms of the disease are still not fully understood, and so far there is neither a cure nor even an effective treatment to delay its onset and progression. A key characteristic of Alzheimer's disease is the formation of insoluble plaques, primarily consisting of amyloid-β (Aβ) peptides, and neurofibrillary tangles in the brain tissue, which triggers neurodegenerative changes leading to the death of neurons. The so-called amyloid hypothesis, put forward thirty years ago, suggests that the accumulation of amyloid plaques is one of the main causes of Alzheimer's disease. In 2018, for the development of this theory and “for groundbreaking research on the genetic and molecular basis of Alzheimer’s disease, with far-reaching implications for the development of new therapeutic interventions as well as for the understanding of other neurodegenerative diseases of the brain” four neuroscientists Bart De Strooper, Michel Goedert, Christian Haass, and John Hardy were awarded the Brain Prize, the world's most prestigious award for brain research.
But what causes amyloid-β aggregation and deposition, what role interactions between peptides and cell membranes may play in this mechanism, and how to prevent the formation of plaques outside the membrane, are still unknown.
Interactions in the membrane: Aß-peptides, cholesterol and melatonin
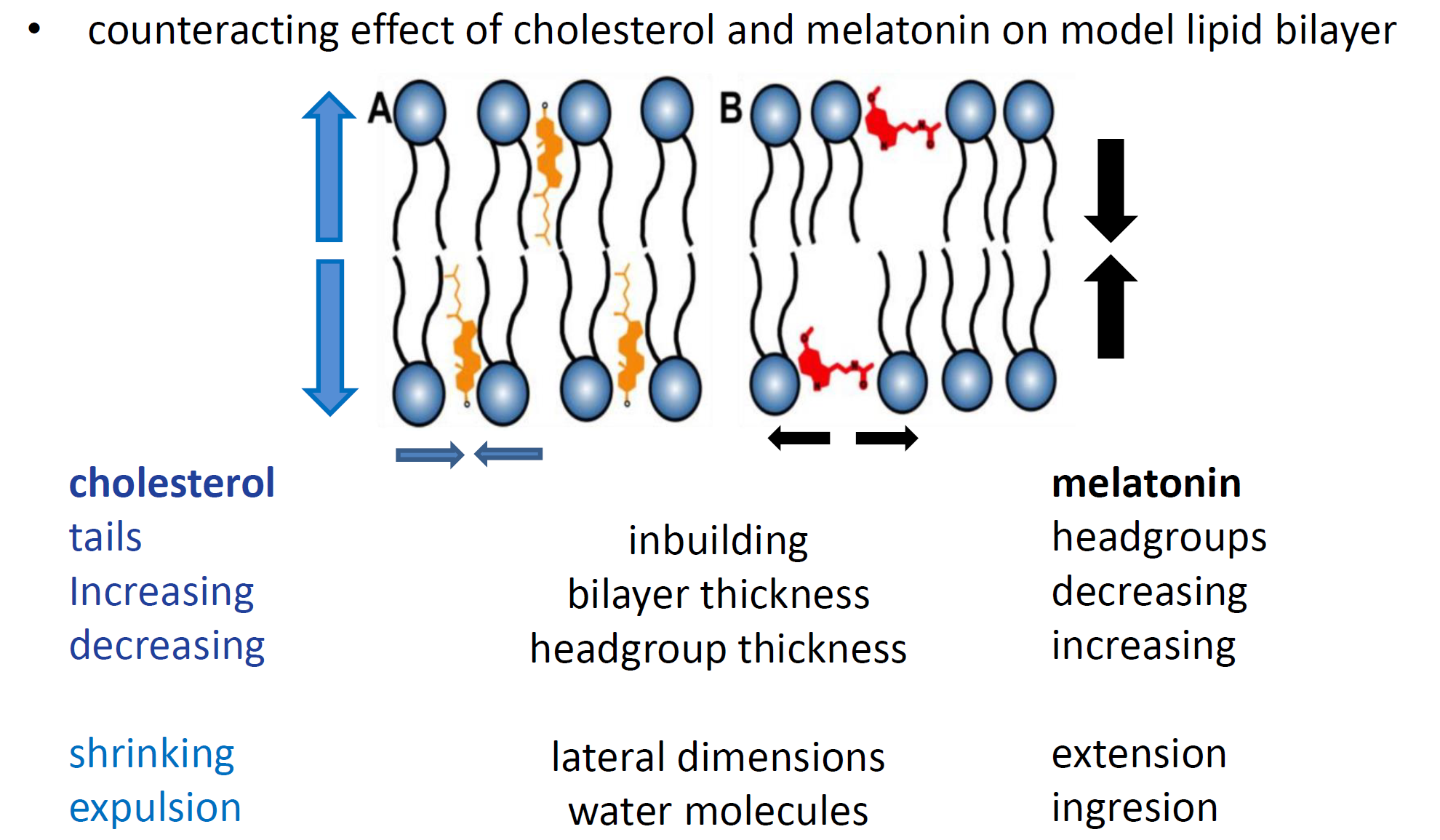
The functioning of the nervous tissue is associated with biomembranes, the structural and metabolic properties of which are determined by numerous lipids that make up more than 50% of the brain’s dry weight. The elasto-mechanical properties of membranes depend on the shape, size and chemical composition of lipids. It is these membrane properties, its chemical composition and thickness that determine the permeability to various substances of the medium. Penetrating into the membrane, they can provoke and aggravate the disease, or, on the contrary, prevent its development. For example, proteins, important components of the membrane, actively transport substances through it. In turn, the permeability and fluidity of the membrane are affected by such substances present in the brain as cholesterol and melatonin, the effect of which scientists are actively investigating. As is known, the level of melatonin in the body decreases and the level of cholesterol rises with age. Alzheimer's disease tends to occur later in life, so the effect of melatonin and cholesterol on the lipid membrane is becoming increasingly important.
Cholesterol, being embedded into the membrane, leads to an increase in the ordering of lipid chains and the overall stiffness of the membrane, reducing the dynamic properties and affecting the transportation of various substances through the membrane. Melatonin, on the contrary, has a completely different effect. It has been shown to increase the fluidity of the membrane and counteract the effect of cholesterol. Thus, the opposite effects of these two molecules naturally suggest their use to regulate the physical properties of membranes and possibly to control the formation of amyloid fibrils and their toxicity. Many functional disorders and various diseases, including Alzheimer's, are associated with changes in membrane fluidity, when for some reasons amyloid beta peptides begin to destroy the membrane of neurons and build up, forming plaques. Perhaps, by finding the cause of this, it will be possible to stop the process of accumulation of peptides or retain them within the membrane interior and prevent the development of the disease?
Armed with methods of small-angle neutron scattering, diffraction and reflectometry, which allow determining the main parameters of membranes, scientists from the Frank Laboratory of Neutron Physics within a research team tried to shed light on the ongoing structural changes and study the processes of accumulation of beta-amyloid peptide in model biological membranes, containing cholesterol and melatonin as agents affecting the fluidity of membranes.
Neutrons and the membrane: experimental results
In the study, the researchers corroborated the stiffening and fluidizing effects of cholesterol and melatonin, respectively, as well as the thickening of cholesterol-loaded membranes and thinning of melatonin-containing ones, and further examined the changes caused by the presence of amyloid beta peptides (Aβ25-35) in the lipid bilayer model membrane.
In this case, the structure of membranes is affected by a change in temperature, the presence of a charge in the medium, an increase in the concentration of ions and their hydration properties.
As shown by neutron scattering experiments, a decrease in the membrane thickness is associated with the incorporation of amyloid beta peptide. With the addition of cholesterol, which increases membrane stiffness, the membrane thickness also decreases due to the incorporation of the peptide. At the same time, the addition of the peptide to melatonin-containing membranes results in only a small change in the membrane thickness. This observation led to the assumption of a different localization of the peptide within the membrane in this case.
“Our experiments revealed differences in the interaction of amyloid beta peptide with the model membranes that contain cholesterol or melatonin. The visualization of molecular dynamics simulations performed together with our colleagues from LIT and LRB JINR show that in the case of a ‘stiff’ cholesterol-containing membrane, the peptide becomes localized at the water-membrane interface, where it interacts with polar lipid head groups. In a ‘fluid’ melatonin-containing membrane, the peptide is incorporated into the hydrophobic core of the membrane. The obtained results point to the key role of membrane viscosity in the peptide incorporation into the membrane. They can be used in future studies of the damaging effects of amyloid and in the search for ways to block or reduce its effects, since they may provide some insight into the molecular mechanism of the protective function of melatonin in Alzheimer's disease,” said one of the study's authors, Norbert Kučerka, FLNP Deputy Director for Research.
Tatiana Murugova, Oleksandr Ivankov, Elena Ermakova, Tomáš Kondela, Pavol Hrubovčák, Vadim Skoi, Alexander Kuklin and Norbert Kučerka. Structural changes introduced by cholesterol and melatonin to the model membranes mimicking preclinical conformational diseases. March 2020. General Physiology and Biophysics. 39(02):135-144. DOI: 10.4149/gpb_2019054
Olga Baklitskaya-Kameneva